Product Description
Mesenchymal stem cells (MSC) derived from human placenta are a well-characterized population of adult stem cells. Number of researchers have shown that adult MSCs have a broad therapeutic potential due to their capability to renew and differentiate into various lineages of mature cells that produce fat, cartilage, bone, tendon, and muscle when cultured under specific permissive conditions [1]. In addition, placenta derived MSC show differentiation capacity toward both osteo-blasts and adipocytes thus serving as a great model for studying the molecular basis of differentiation [2]. These properties, in combination with their developmental plasticity, have generated tremendous interest in regenerative medicine to replace damaged tissues. These findings have spurred the development of MSC-based therapies for treating wide range of non-skeletal diseases [3].
iXCells Biotechnologies provides placental MSC isolated from different layers of human placenta, including decidua (Cat# 10HU-170), chorionic villi (Cat# 10HU-171), and chorionic plate (Cat# 10HU-172). Each vial contains ≥ 0.5 million cells. These cells are expanded in Mesenchymal Stem Cell Medium (Cat# MD-0037) and then cryopreserved at passage 2. The human placental MSC express typical mesenchymal cell surface markers, such as CD105, CD73, and CD90 and are negative for hematopoietic markers including CD34, CD45 and endothelial cell marker CD31 [Figure 1, 2]. These cells can further be differentiated into adipocytes using Adipocyte Differentiation Medium (Cat# MD-0005) and into osteoblasts using Osteogenic Differentiation Medium (Cat# MD-0006) [Figure 3 and Figure 4]. Human placental MSC are negative for mycoplasma, bacteria, yeast, and fungi and can be expanded for no more than 3 passages in iXCells’ Mesenchymal Stem Cell Medium.

Figure 1. Top: Phase contrast images of human placental mesenchymal stem cell-Decidua (Cat# 10HU-170) taken at day 1 and day 3 post-recovery. Bottom: ICC staining using antibodies against CD73 (Green) and CD90 (Green), separately.
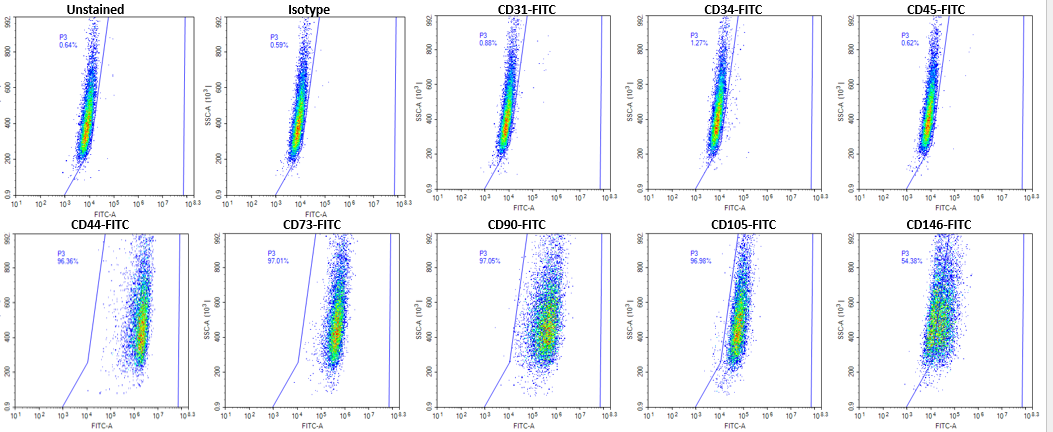
Figure 2. The purity of human placental mesenchymal stem cells-Decidua (Cat# 10HU-170) was determined using the corresponding antibodies by flow cytometry analysis. The sample stained with isotype control was used for gating strategy.
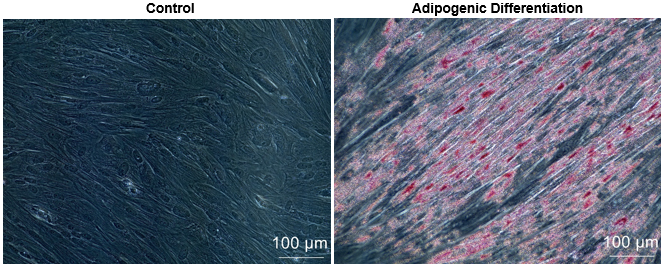
Figure 3. Human placental mesenchymal stem cells-Decidua (Cat# 10HU-170) adipocyte differentiation (Day 22 post adipocyte induction).
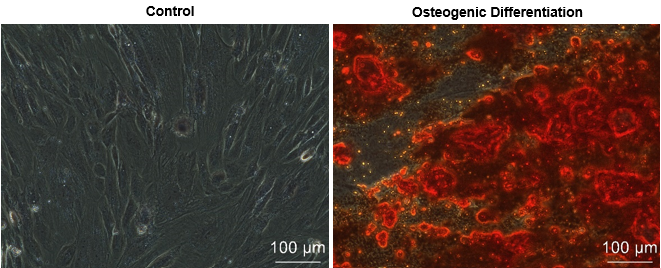
Figure 4. Osteogenic differentiation (Day 20 post osteocyte induction) using human placental mesenchymal stem cells -Decidua (Cat# 10HU-170). Alizarin Red S staining of osteoblasts. The extracellular calcium deposit was stained in bright orange-red color.
Product Details
| Tissue | Human Placenta |
| Package Size | 0.5 million cells/vial |
| Passage Number | P2 |
| Shipped | Cryopreserved |
| Storage | Liquid nitrogen |
| Growth Properties | Adherent |
| Media | Mesenchymal Stem Cell Medium (Cat# MD-0037) Adipocyte Differentiation Medium (Cat# MD-0005) Osteogenic Differentiation Medium (Cat# MD-0006) |
References
[1] Miao, Z., Jin, J., Chen, L., Zhu, J., Huang, W., Zhao, J., Qian, H., & Zhang, X. (2006). Isolation of mesenchymal stem cells from human placenta: comparison with human bone marrow mesenchymal stem cells. Cell biology international, 30(9), 681–687.
[2] Fukuchi, Y., Nakajima, H., Sugiyama, D., Hirose, I., Kitamura, T., & Tsuji, K. (2004). Human placenta-derived cells have mesenchymal stem/progenitor cell potential. Stem cells (Dayton, Ohio), 22(5), 649–658.
[3] García-Gómez I, Elvira G, Zapata AG, Lamana ML, Ramírez M, Castro JG, Arranz MG, Vicente A, Bueren J, García-Olmo D.(2010) Mesenchymal stem cells: biological properties and clinical applications. Expert Opin Biol Ther. 10(10):1453-68.
Starting at: $847.00
Starting at: $847.00
FREQUENTLY BOUGHT TOGETHER - RECOMMENDED FOR YOU!